Types of Covid-19 Vaccine and their Differences
Vaccines prompt an immune response so that your body remembers how to fight a virus in the future. Some vaccines use a whole virus to cause your immune system to respond. Other vaccines use parts of the virus or genetic material that provides instructions for making specific proteins like those in the virus.

A coronavirus disease 2019 (COVID-19) vaccine can help you develop immunity to SARS-CoV-2, the virus that causes COVID-19, without getting ill. Vaccines prompt an immune response so that your body remembers how to fight a virus in the future. Some vaccines use a whole virus to cause your immune system to respond. Other vaccines use parts of the virus or genetic material that provides instructions for making specific proteins like those in the virus.
How do vaccines work?
All vaccines work by teaching our bodies to recognise and fight the pathogen in a safe way. They encourage our immune system to produce antibodies, T-cells or both, so that if we encounter the infection later our immune system knows how to defend against it.
Vaccines can be made using many different technologies. The Covid-19 vaccines that are currently the most advanced are using four different approaches:
- Viral Vector Vaccine
- Genetic Vaccines(RNA)
- 'Whole' Virus Vaccine
- Protein Subunit Vaccine
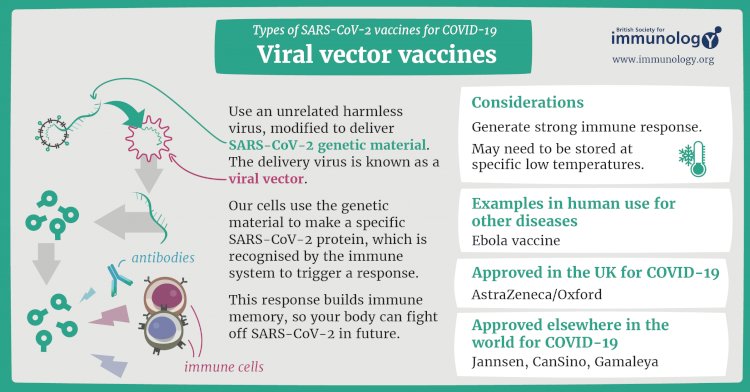
Viral vector vaccines for COVID-19
This type of vaccine uses an unrelated harmless virus (the viral vector) to deliver SARS-CoV-2 genetic material. When administered, our cells use the genetic material to produce a specific viral protein, which is recognised by our immune system and triggers a response. This response builds immune memory, so your body can fight off the virus in future.
Oxford-AstraZeneca is the first viral vector vaccine to be approved for Covid-19. More are in late-stage research, such as CanSino Biologics, Gamaleya Research Institute and Johnson & Johnson. They all use adenoviruses – a group of viruses that cause the common cold – as the vector or carrier.
Another example of a viral vector vaccine is the Ervebo (rVSV-ZEBOV) Ebola vaccine – it uses the vesicular stomatitis virus as the carrier.
Vaccines include: Oxford-AstraZeneca, Sputnik V (Gamaleya Research Institute) vaccine uses this technology to protect against COVID-19.
Benefits: Viral vector-based vaccination is another well-established technology that can trigger a strong immune response as it also involves both B cells and T cells.
Challenges: Previous exposure to the vector could reduce effectiveness, plus these types of vaccines are relatively complex to manufacture compared to others.
Other licensed vaccines that use this type of technology: Ebola
Credits To : www.immunology.org
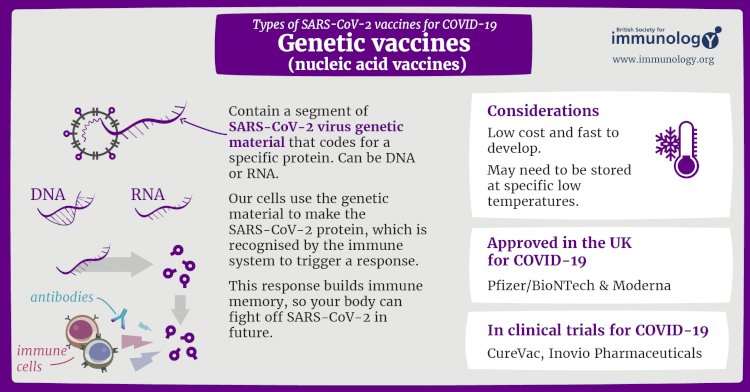
Genetic vaccines for COVID-19
Unlike vaccine approaches that use either a weakened or dead whole microbe or parts of one, a nucleic acid vaccine just uses a section of genetic material that provides the instructions for specific proteins, not the whole microbe. DNA and RNA are the instructions our cells use to make proteins. In our cells, DNA is first turned into messenger RNA, which is then used as the blueprint to make specific proteins.
A nucleic acid vaccine delivers a specific set of instructions to our cells, either as DNA or mRNA, for them to make the specific protein that we want our immune system to recognize and respond to.
The nucleic acid approach is a new way of developing vaccines. Before the COVID-19 pandemic, none had yet been through the full approvals process for use in humans, though some DNA vaccines, including for particular cancers, were undergoing human trials. Because of the pandemic, research in this area has progressed very fast and some mRNA vaccines for COVID-19 are getting emergency use authorization, which means they can now be given to people beyond using them only in clinical trials.
Vaccines include: Pfizer-BioNTech, Moderna are Two RNA Covid-19 vaccines that have been approved for use Both have reported high levels of vaccine efficacy – around 95%.
Benefits: According to the University of Cambridge’s PHG Foundation, advantages include good safety (since there are no live components, there’s no risk of the vaccine triggering disease), reliability, and that it’s relatively simple to manufacture.
Challenges: Disadvantages include unintended effects (such as an unintended immune reaction), ensuring effective delivery into the body (since free RNA in the body is quickly broken down), storage issues, plus the fact that this type of vaccine has never previously been licensed for humans.
Other licensed vaccines that use this type of technology: None
Credits To : www.immunology.org
The whole-microbe approach
Inactivated vaccine
The first way to make a vaccine is to take the disease-carrying virus or bacterium, or one very similar to it, and inactivate or kill it using chemicals, heat or radiation. This approach uses technology that’s been proven to work in people – this is the way the flu and polio vaccines are made – and vaccines can be manufactured on a reasonable scale.
However, it requires special laboratory facilities to grow the virus or bacterium safely, can have a relatively long production time, and will likely require two or three doses to be administered.
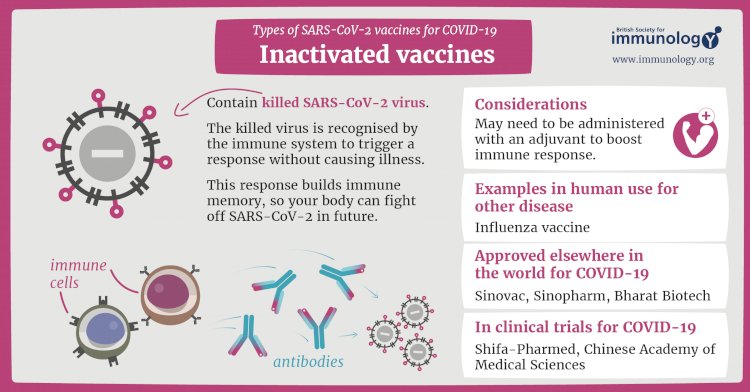
Credits To : www.immunology.org
Live-attenuated vaccine
A live-attenuated vaccine uses a living but weakened version of the virus or one that’s very similar. The measles, mumps and rubella (MMR) vaccine and the chickenpox and shingles vaccine are examples of this type of vaccine. This approach uses similar technology to the inactivated vaccine and can be manufactured at scale. However, vaccines like this may not be suitable for people with compromised immune systems.
One vaccine in phase III clinical trials is virus-like particle: Medicago Inc. An example of an existing vaccine is the HPV/cervical cancer vaccine.
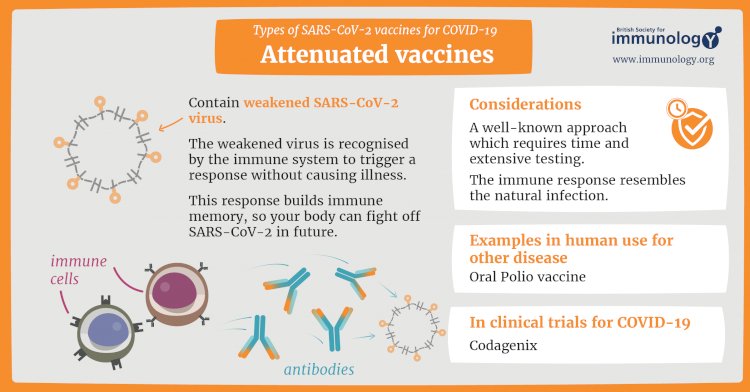
Credits To : www.immunology.org
Vaccines include: Sinopharm, Sinovac
Benefits: According to Gavi, the Vaccine Alliance (GAVI), the advantages of an inactivated whole virus vaccine include the fact its technology is well established, it is suitable for people with compromised immune systems, and it’s relatively simple to manufacture.
Challenges: Booster shots may be required.
Other licensed vaccines that use this type of technology: Hepatitis A, polio, rabies (all inactivated type)
Protein subunit vaccines
A subunit vaccine is one that only uses the very specific parts (the subunits) of a virus or bacterium that the immune system needs to recognize. It doesn't contain the whole microbe or use a safe virus as a vector. The subunits may be proteins or sugars. Most of the vaccines on the childhood schedule are subunit vaccines, protecting people from diseases such as whooping cough, tetanus, diphtheria and meningococcal meningitis.
Cells like this act as factories, building large quantities of the protein – which is then extracted, purified and used as the active ingredient in the vaccine.
When it is injected, our bodies learn to recognise the viral protein so that they can mount an immune response which protects against future infection.
An example of an existing protein subunit vaccine is for hepatitis B, which uses yeast cells to build the virus protein.
Vaccines include: Novavax and Chinese Academy of Sciences.
Benefits: The protein subunit vaccination is also a well-established technology that’s advantageous for those with compromised immune systems.
Challenges: This type of vaccine is relatively complex to manufacture, and adjuvants and booster shots may be required.
Other licensed vaccines that use this type of technology: Hepatitis B, meningococcal disease, pneumococcal disease, shingles
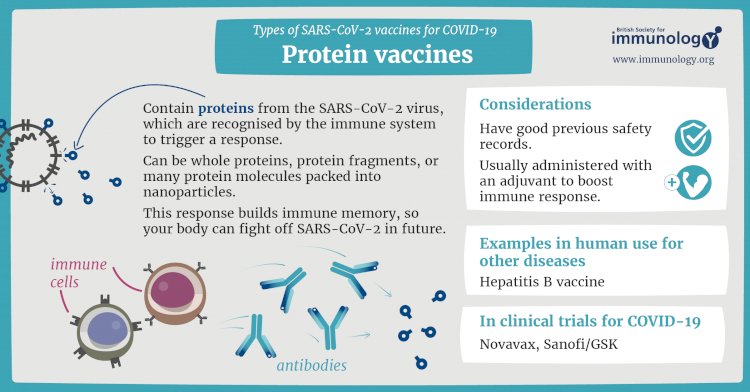
Credits To : www.immunology.org
Covishield
Covishield is also known as s ChAdOx1 nCoV-19, or AZD1222, developed by Oxford University in partnership with AstraZeneca. Its manufacturing and trial partner is the Serum Institute of India, Pune, and ICMR.
This vaccine uses a replication-deficient chimpanzee viral vector based on a weakened version of adenovirus that causes infections in chimpanzees and consists of the genetic material of the coronavirus spike protein, which helps the virus to bind with the human cells. The modified chimpanzee adenovirus can’t replicate, hence do not cause infection, and rather serves as a vector to transfer the coronavirus spike protein.
Like most of the vaccines, the Covishield produces the mimic spike protein that causes an immunological response, which would finally prime the immune system.
Covishield is approved in India, Argentina, El Salvador, Mexico, Bangladesh, and the Dominican Republic regulatory authorities for emergency use.
Covaxin
Covaxin is India’s first indigenous COVID-19 vaccine. It is an inactivated vaccine that suppresses the virus’ capability to duplicate yet keeps it unimpaired so that the immune system can still recognize it and create an immune reaction. Covaxin helps in increasing the production of antibodies in the host body.
This vaccine can target the new variant of SARS-CoV-2 that was recently detected in the UK. This vaccine produces a strong immune response and in-vitro viral neutralization. Both homologous (vaccine virus strain) and heterologous (divergent) SARS-CoV-2 strains were neutralized using this vaccine, as per the vaccine-induced antibody responses recorded.
After successfully finishing the interim report from the Phase 1 & 2 clinical trials of COVAXINTM, it was approved by the Drugs Controller General of India for Phase 3 clinical trials in 26,000 participants in over 25 centers across India.

Sputnik V
Sputnik V is the globe’s first registered COVID-19 vaccine based on a viral two-vector vaccine based on two human adenoviruses. The vaccine is manufactured in Russia but is approved for use in India and is being imported. Sputnik V is also known as Gam-COVID-Vac. Sputnik V vaccine is titled after the 1st Soviet space satellite.
Clinical trials of Sputnik V have been introduced in India, Venezuela, Belarus, and the UAE, and RDIF’s global partners in India, Brazil, China, South Korea, and other countries will manufacture the vaccine for the international market.
Gennova Biopharmaceuticals
Gennova Biopharmaceuticals, in partnership with HDT Biotech Corporation, has actually collaborated to establish an mRNA vaccine. Their vaccine is referred to as HGCO19, has shown safety, immunogenicity, neutralization antibody activity in animal models.
mRNA vaccine candidate HGCO19 has a short, synthetic version encoding the spike protein of SARS-CoV-2. When the vaccine is injected into the body, the synthetic mRNA is taken to muscle cells, where it guides cells to make many copies of mRNA and copies of the antigen.
SARS-CoV-2 also utilizes its RNA genome led method to expand and express viral proteins inside the host cells.
Mynvax
Mynvax’s COVID-19 vaccine uses a unique method involving a protein-based vaccine. This vaccine can be stored at room temperature for 1 month without losing its effectiveness. This eliminates the need for cold chains. The vaccine is now in the phase for formal safety and toxicity studies, process development, human clinical trials, and manufacturing.
This heat-tolerant vaccine can endure 70º C for nearly 16 hours, and the freeze-dried form of the vaccine can withstand 100º C.
This vaccine candidate includes a part of the spike protein of SARS-CoV-2, known as the receptor-binding domain (RBD).
Biological E
Biological E has started Phase I/II clinical trials to assess the safety and immunogenicity of its COVID-19 subunit vaccine candidate after it was approved by DGCI.
The clinical trials will also analyze the capability to generate an immune response of the vaccine candidate, which has the Receptor Binding Domain of the Spike Protein of SARS-CoV-2 at three dose levels adjuvanted with CpG 1018 plus alum.
The human trials will include 360 healthy individuals of 18-65 years. The vaccine will be given as two dosages 28 days apart.
The ICMR, in partnership with Biological E Ltd, developed “highly purified antisera” developed by injecting inactivated SARS-CoV2 in horses, which can be used for treating prophylaxis and COVID-19.
Zydus Cadila
Zydus Cadila, Ahmedabad ZyCoV-D established the DNA vaccine system in India, known to show enhanced vaccine stability, demanding lower cold chain requirements, making the vaccine ideal for access to remote areas of the nation.
Zydus Cadila received approvals from the DCGI to initiate Phase III Clinical Trial of ZyCoV-D.
The manufacturing process is easier while using this platform, and the platform can be altered in just a few weeks if the virus gets mutated.
The plasmid DNA, when introduced into the host cells, would be translated into the viral protein and will evoke a robust immune reaction, which helps in protecting from COVID-19 and viral clearance. As per phase I and II clinical trials, ZyCoV-D is safe, immunogenic, and well-tolerated.
|
Basis of distinction |
Covishield |
Covaxine |
Sputnik V |
ZyCoV-D |
HGCO19 – Gennova Biopharmaceuticals |
Mynvax |
Biological E |
|
Developed By |
University of Oxford and AstraZeneca in collaboration with the SII |
Bharat Biotech in association with ICMR and NIV |
Gamaleya Research Institute of Epidemiology and Microbiology, Russia. (Approved to be used in India) |
Zydus Cadila |
Gennova Biopharmaceuticals |
Mynvax -IISc |
Biological E in collaboration with the J&J. |
|
Vaccine Type |
Modified chimpanzee adenovirus vector |
Inactivated whole virus |
Non-replicating viral vector (adenovirus) |
Plasmid DNA vaccine |
mRNA vaccine |
Protien based vaccine |
Subunit vaccine |
|
Efficacy |
DCGI: 70.42% overall |
78% |
91.4% |
N/A |
N/A |
N/A |
66% |
|
Storage Temperature |
2-8°C |
2-8°C |
−18 °C |
2-8°C |
2-8°C |
37º C |
N/A |
|
Dosage |
Two doses (Gap 2.5-3 Months) |
Two Doses (0, 14 Days) |
Two dose(21 days) |
Two dose |
N/A |
N/A |
Two dose (28 Days gap) |
|
Routes of administration |
Intramuscular injection |
Intramuscular injection |
Intramuscular injection |
Intramuscular injection |
N/A |
N/A |
Intramuscular injection |
(The Article has been taken from various syndicate platform.)
[Credits to : www.healthcareitnews.com , www.who.int , www.wellcome.org , www.immunology.org , www.biotecnika.org ]

 Arindam Deshmukh
Arindam Deshmukh